| Literature link | GPT Summary | Evidence category | Disease type |
|---|---|---|---|
| 21738950 | Calprotectin, a novel marker of obesity, has recently gained attention for its potential role in insulin resistance and inflammation. This study aimed to evaluate the circulating levels and tissue expression of calprotectin subunits (S100A8 and S100A9) in visceral adipose tissue (VAT) and investigate their impact on obesity, insulin resistance, and inflammation, as well as the effects of weight loss. The study included 53 subjects, and gene expression levels of the S100A8/A9 complex were analyzed in VAT, adipocytes, and stromal vascular fraction cells (SVFCs). Circulating levels of calprotectin and soluble receptor for advanced glycation end products (sRAGE) were measured before and after weight loss through Roux-en-Y gastric bypass (RYGB) (n = 26). The results showed that circulating calprotectin levels and VAT expression of the S100A8/A9 complex were significantly higher in both normoglycemic and type 2 diabetic obese patients, with a strong correlation to inflammatory markers. In contrast, sRAGE levels were significantly lower in obese groups compared to lean controls. Following RYGB, calprotectin levels decreased significantly, while sRAGE levels showed a trend toward increase. Expression of calprotectin was primarily found in SVFCs, with significant correlation to the mRNA levels of monocyte-macrophage-related molecules, including CD68, MCP1, CD11B, and NOX2. Furthermore, TNF-α treatment increased S100A8 expression in human visceral adipocytes. The elevated levels of calprotectin in obesity and type 2 diabetes, along with its association with inflammation and higher expression in SVFCs, suggest that calprotectin may act as a chemotactic factor, contributing to macrophage recruitment in VAT and exacerbating inflammation, which may drive obesity-associated comorbidities. |
Mechanism |
Obesity |
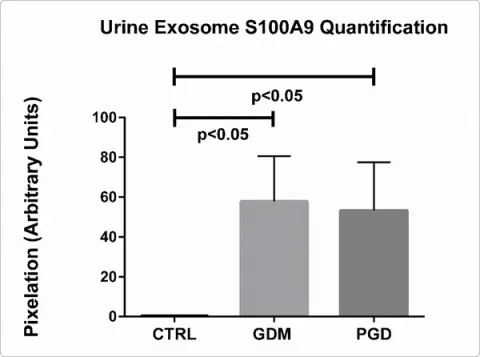
| 31448371 | This research aimed to explore the differences in the exosomal proteome of 24-hour urine samples collected from pregnant women with GDM, PGD, and controls at 20 weeks of pregnancy. The study identified an increase in S100A9 protein in GDM and PGD subjects, which was linked to maternal obesity and macrosomia in newborns. The findings suggest that urinary exosome proteomics can provide non-invasive insights into the pathophysiology of diabetes during pregnancy and help differentiate between types of diabetes on a pathway level. | Risk factor | GDM |
| 31363816 | Obesity is a widespread condition closely linked to various comorbidities, including hypercholesterolemia, hypertension, cardiovascular diseases, type 2 diabetes, and cancer. Adipose tissue in obese individuals exhibits hypertrophic adipocytes, adipocyte hyperplasia, and chronic low-grade inflammation. S100 proteins, a group of Ca2+-binding proteins expressed specifically in various cell types, have been implicated in regulating multiple cellular functions. These proteins act as intracellular Ca2+ sensors and transduce signals from extracellular factors through interaction with a range of membrane receptors. Notably, S100A4, the S100A8/S100A9 heterodimer, and S100B have been associated with macrophage-driven inflammation in obesity, mediated through toll-like receptor 4 and receptor for advanced glycation end-products (RAGE) signaling. Elevated levels of S100A4, S100A8/S100A9, S100A12, and S100B in serum correlate with insulin resistance, metabolic risk, and adipocyte size. Interestingly, secreted S100B also appears to have neurotrophic effects on sympathetic fibers in brown adipose tissue, contributing to enhanced sympathetic innervation compared to white adipose tissue. This review provides an overview of S100 proteins and critically explores their roles in the pathophysiology of obesity. | Mechanism | Obesity |
| 23709000 | The study investigates the impact of gestational diabetes mellitus (GDM) on the proteome of maternal omental adipose tissue using 2D-DIGE and mass spectrometry. It identifies 14 differentially expressed proteins, including up-regulation of COL6A2, FIBB, LUM, and S100A9, and down-regulation of 10 others, highlighting the involvement of these proteins in inflammation, lipid and glucose metabolism, and oxidative stress. The findings support the role of visceral adiposity in the pathogenesis of GDM. | Risk factor | GDM |
| KEGG pathway |
|---|
| IL-17 signaling pathway |
RF's name
S100A9
RF's type
mRNA